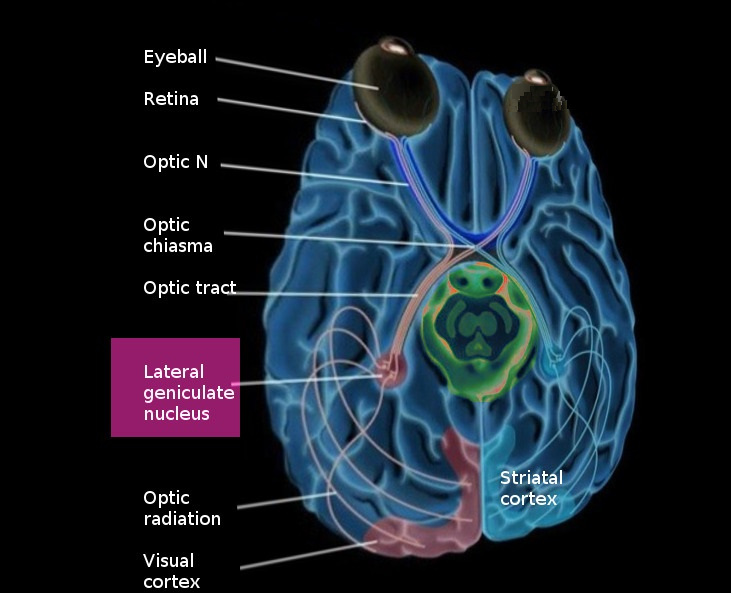
The lateral geniculate nucleus (LGN) is a tiny area in the back of the thalamus. Nerve fibers from the retinal ganglion cells enter the lateral geniculate nucleus of the thalamus from the optic chiasm. These fibers constitute only a few percent of the inputs to the LGN. There are other inputs, some from the superior colliculus, some from other areas of the thalamus, but most from the visual cortex. Apparently the input from the retina is controlled in many ways before it reaches the primary visual cortex.
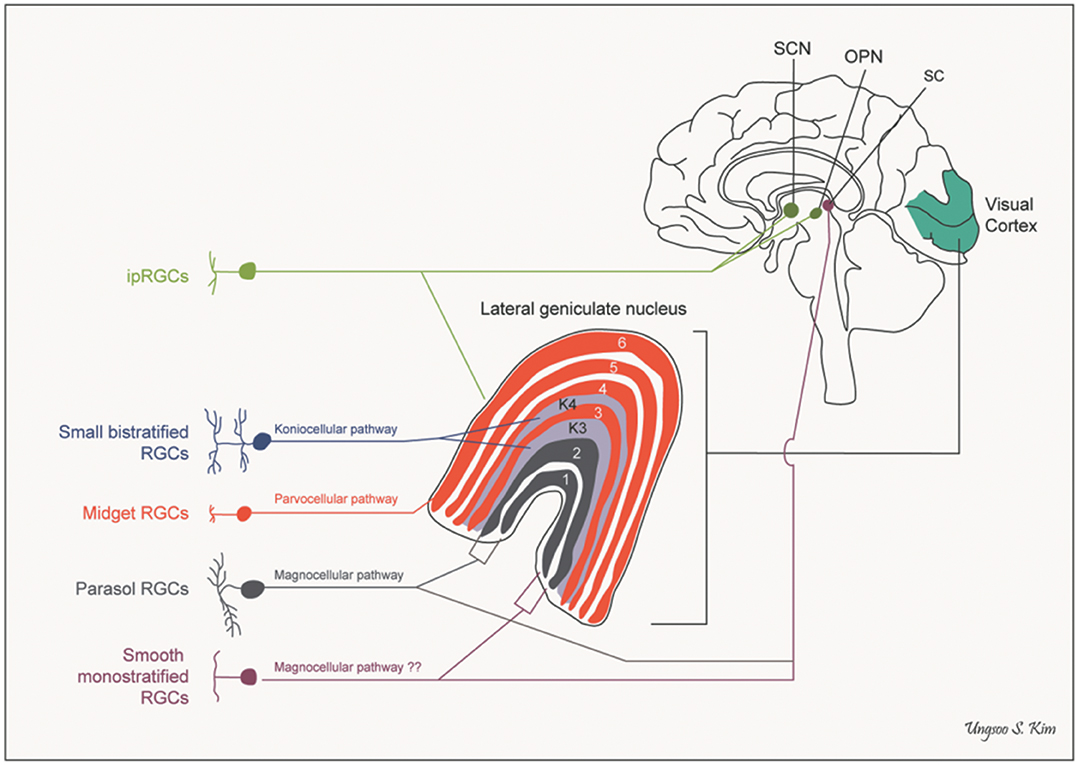
The fibers entering the LGN cross at the level of the optic chiasm, and by the time they get to the visual cortex the right side of the brain ends up looking at the left half of the visual field. However the inputs from each eye are kept segregated in the layers of the LGN. In humans there are 6 layers, three of them (2,3,5) get input from the same side and three of them (1,4,6) get input from the opposite side. The magnocellular M-type neurons (low contrast, high speed) project to layers 1 and 2, and the parvocellular P-type neurons (high contrast, low speed) project to 3 through 6. In additional the koniocellular K-type cells (which subserve specific kinds of color opponency like yellow-blue) project to areas just ventral to each of the other layers. Every set of connections maintains the retinal topography.
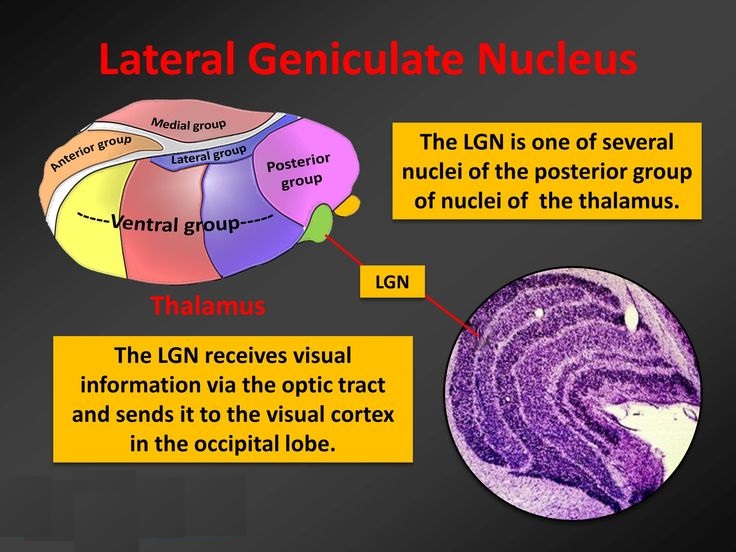
The lateral geniculate nucleus (LGN) is a tiny area in the back of the thalamus, consisting of six very prominent stainable layers under the light microscope, with some additional small layers sandwiched between them. The layers are specific to different kinds of connections from both retinas, ipsilateral and contralateral.
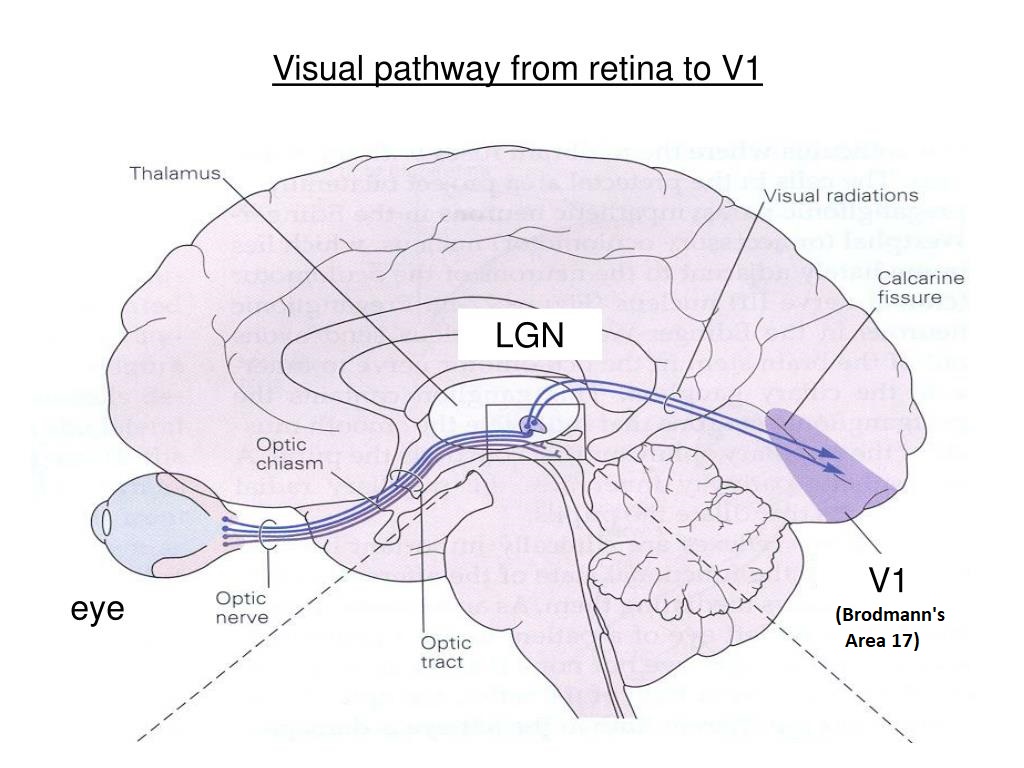
Axons from thalamo-cortical relay neurons in the LGN travel into the posterior occipital lobe where they connect topographically with specific layers of the primary visual cortex (V1).
The LGN has long been considered as a "way station" between the retina and the primary visual cortex, but it's much more than that. The cerebral cortex engages in several forms of predictive coding (which we'll talk about in considerable detail in subsequent sections), one of which is that it fills in the blind spot during perception. Once an object has been identified by the brain, and once it is being tracked through the visual field and across the timeline, the signals arriving from the retina need to be compared with predictions made in advance. The importance of downstream information flow can be understood by comparing the relative amounts of input to the LGN. Retinal inputs constitute less than 10% of the LGN input, the rest comes from the cerebral cortex (about 50% from layer 6 neurons in the primary visual cortex) and from the brainstem and other thalamic areas. The neuronal firing dynamics are important to understand as well, as is the synchronization of visual activity with eye movements and with population dynamics like the alpha rhythm. While the retina may emphasize spatial decorrelation through its center-surround organization, the LGN emphasizes temporal decorrelation through its internal connections and its connections with the cerebral cortex. The figure schematizes some of the pathways through the LGN.
LGN CircuitryRetino-geniculate axons are excitatory, the ganglion cells use glutamate as a neurotransmitter. The axons entering the LGN branch sparsely into one or more clusters, typically an LGN relay neuron will receive inputs from just a few retinal ganglion cells. However the LGN receptive field are larger and more complex, as we'll see below. Synapses from the retina typically occur on the proximal dendrites and dendritic shaft segments of the relay cells, which means they drive the relay neurons reliably, close to the site of action potential generation in the axon hillock. The peripheral dendrites of the relay cells get inputs from cortical feedback neurons originating in layer 6 of the primary visual cortex, and interneurons from both inside and outside the thalamus (like from the thalamic reticular nucleus).
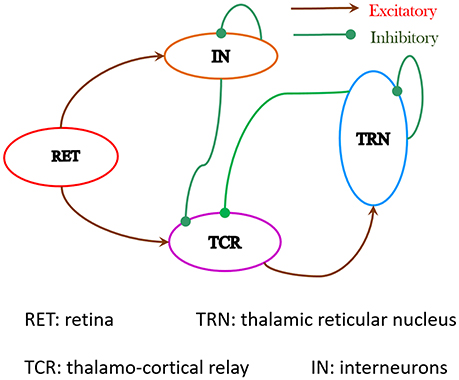
About 3/4 of the neurons in the LGN are thalamo-cortical relay neurons. The remainder are interneurons, mostly inhibitory using GABA as a neurotransmitter. The interneurons provide feed-forward inhibition to the relay cells, which occurs in the parts of the interneuron peripheral dendrites that engage in complex synapses with the incoming fibers from the retina. The range of response patterns for interneurons includes initial sags, rebound bursts, tonic firing, spike time adaptation, and periodic bursting. The interneurons are also important for synchronizing thalamic oscillations (Halnes et al 2011).
While the circuitry of the LGN is relatively simple, it is important to understand that the input from the retina is being combined in different ways into the thalamo-cortical input. For example inactivation of On-type bipolar cells in the retina does not inactivate all of the On-type responses in the LGN, instead 50% of the On-type LGN neurons rapidly develop Off-type responses (B Moore et al 2011). The LGN is far from a simple relay, there is considerable reorganization of the information from the retinal channels, and there is the introduction of additional dynamics.
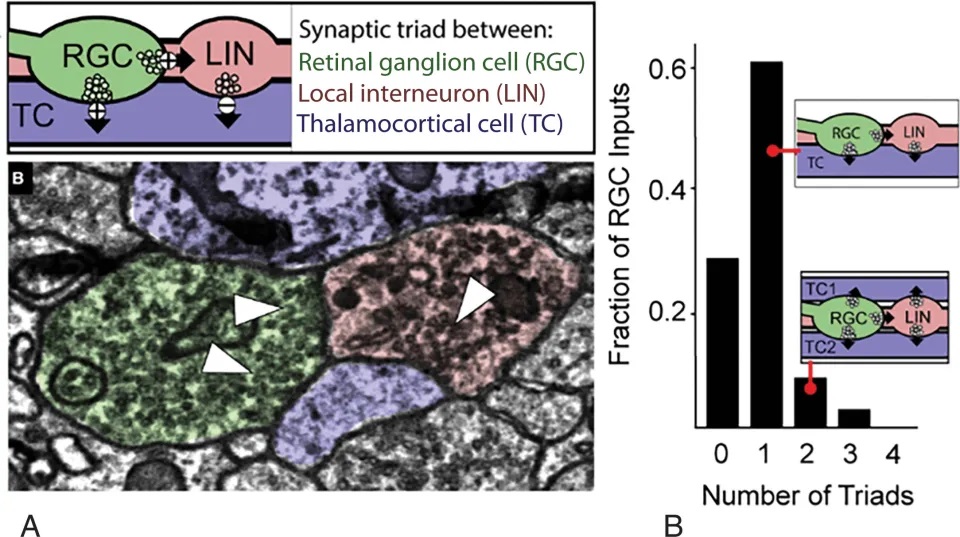
Retino-geniculate synapses are unusual. Retinal axon terminals synapse near the cell bodies of retinal ganglion cells, onto the dendrites and dendritic shafts. Typically there are as many as 50 synapses between an incoming fiber and its target relay neuron. Some of the retinal synapses look pretty ordinary, while others are encapsulated in large glomeruli containing multiple inputs from local inhibition, cortical feedback, and neuromodulatory systems like the raphe and locus coeruleus. The figure shows one of the triadic synapses in the LGN.
Inhibitory interneurons in the LGN receive direct inputs from the retina. They inhibit relay neurons, and they inhibit themselves. The self inhibition behavior does two things: first it tends to let through early activity while damping later activity. And second, the delays inherent in mutual inhibition can generate filters that ring and networks that oscillate.
As with most thalamo-cortical relay cells (Weyand et al 2001), LGN relay neurons have two modes: tonic and bursting (Sanchez et al 2023). Bursts are typically preceded by lengthy inter-spike intervals, on the order of 100 milliseconds, but during the burst the spike intervals may shorten to 4 msec or less (Sanchez et al 2023). The receptive fields of burst spikes are different from those of tonic spikes (Alitto et al 2005), indicating that the information being transmitted is different. Compared to tonic spikes, burst spikes have a greater reliability, show a shorter latency from stimulus onset, show a greater dependence on transitions from suppressed to preferred states, and prefer stimuli that strongly drive both the center and the surround of the receptive field.
Both P and M type LGN cells respond with high frequency bursts to strong visual stimulation (Self et al 2005). The P type cell responses in the dorsal layers show center-surround color opponency and the M type responses in the ventral layers prefer high temporal frequencies. Both types of bursting responses are gated during eye closure.
In addition to the projection to the primary visual cortex, the LGN also sends a small number of axons into extrastriate cortex, especially V2 and area MT (V5, which is related to the processing of visual motion). These axons can reorganize themselves and proliferate when V1 is damaged (Atapour et al 2022).
LGN Receptive FieldsA "receptive field" is a loose concept intended to describe the part of ongoing visual activity that the neuron responds to. For example the receptive fields of most retinal bipolar cells are point-like and round, with inhibitory surrounds. But the shape of these receptive fields can change over time, and they can be modulated and controlled in a number of ways, turned on and off, and endowed with a rich variety of time-dependent responses. The general picture that emerges is one of multiple channels processing combinations of luminance, contrast, and transients. The filtering for each channel can change both under local control, and in response to changing stimulus and light conditions.
The receptive fields of retinal photoreceptors are typically very small, in the human central foveal region they may cover a few seconds of arc, extending to a minute or so more peripherally in the fovea. Whereas, receptive fields in the LGN tend to be larger, as if multiple retinal receptive fields are being combined. Nevertheless, the total number of neurons in each LGN is approximately 6 million, corresponding to the number of inputs times the number of layers. This indicates the incoming channels are being recombined before they reach the visual cortex.
LGN receptive fields are tuned in both space and time. The center portion of LGN receptive fields is initially large following a photic stimulus, but shrinks rapidly if the stimulus is maintained (Einevoll et al 2011). Spatial resolution is related to the size of the receptive field center, indicating that the resolution is going from coarse to fine after a photic stimulus. Some of these dynamic changes are related to circuitry inside the LGN itself, while other aspects depend on feedback from the cerebral cortex. The time course of these dynamics is important to understand. For example the initial shrinkage of the center occurs in the first 100 msec, while there is typically a 200-500 msec distance between saccadic eye movements, and therefore the dynamics are likely to be functionally significant.
Here is another area where modeling can help us. To function in this manner the time constants must fall within a certain range, they can be predicted. The time constants relative to network behavior do not necessarily correspond to the time constants at the chemical level, although in many cases there is mutual influence. For example the synaptic time constants can determine the filtering behavior in the channels, and in turn can affect biochemical activity via second messengers. It is extraordinarily difficult to measure and map out the changing receptive fields in living humans. Whereas, a computational model that's even "in the ballpark" can predict important behavior that can be specifically included in subsequent experiments.
Dynamics of LGN NeuronsRetinal neurons fire more than LGN neurons do. Typically LGN relay neurons require convergence of two or more input signals within a 40 msec windows, to generate an action potential. In real life visual conditions, for example when scanning a scene in broad daylight, the visual input is strong and LGN neurons fire reliably in response to retinal input, often achieving the theoretical limit of minimum variance between input and output (Rathbun et al 2010).
The visual signals through the LGN can be modified by other brain areas. For example visual signals tend to be suppressed during saccades. However it is important to bear in mind that the LGN is not the only visual pathway into the cerebral cortex. For example there is a pathway from the superior colliculus into the pulvinar of the thalamus, that's involved in visual modulation relative to attention, and may also be involved in vague perceptions of peripheral motion during blindsight.
Neurons in the lateral geniculate nucleus that project into the primary visual cortex are spiny, and they exhibit calcium driven plateau potentials that are related to bursting behavior. This type of signaling engages time-to-first-spike transmission, which usually indicates hot spots in the visual field. Axons from the retina project onto both excitatory and inhibitory neurons in the LGN, and the inhibitory neurons in turn synapse back onto the excitatory neurons. One of the primary functions of both the retina and the LGN is gain control. Contrast gain control is necessary to prevent the visual pathways from being overloaded by bright light, or understimulated in the dark. LGN neurons respond best to contrast. This is true for gratings as well as for patterns of dots, and movies and real-life visual scenes.
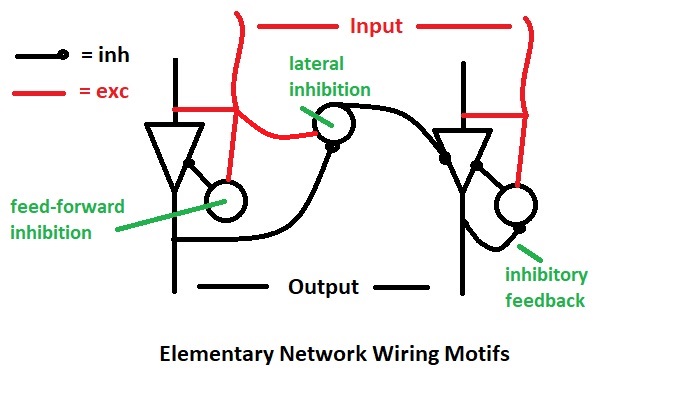
The LGN wiring diagram is an example of basic connection motifs that exist throughout the brain. (Here the word "motif" is used in a different context than that related to information graphs - see Wegner 2024 and Zambra et al 2019). In the periphery, like in the retina and thalamus, these motifs tend to be pretty simple, but in the cerebral cortex they get immensely complicated. From a neural network standpoint, the LGN motifs include local and lateral inhibition. A good modeling tool will have libraries of such motifs, from which intricate and more complex networks can be constructed.
One of the interesting questions is the transmission of wave-like activity from the retina, through the LGN and into the visual cortex. The retinal shift effect is not necessarily "perceived" (although it can be, under contrived conditions), indicating that it is somehow suppressed in subsequent processing. Whether this happens in the LGN is an open question. Again, another area where modeling can help us.
From the LGN, the visual signal travels to the primary visual cortex via the optic radiation. The visual cortex is an analytic engine, it further extracts from the binocular visual signal information related to edge orientation, spatial frequency, and binocular disparity (for depth perception, to identify the boundaries and curvatures of surfaces).
Next: Visual Cortex |






